The Galleri Test

The Galleri® multi-cancer early detection test
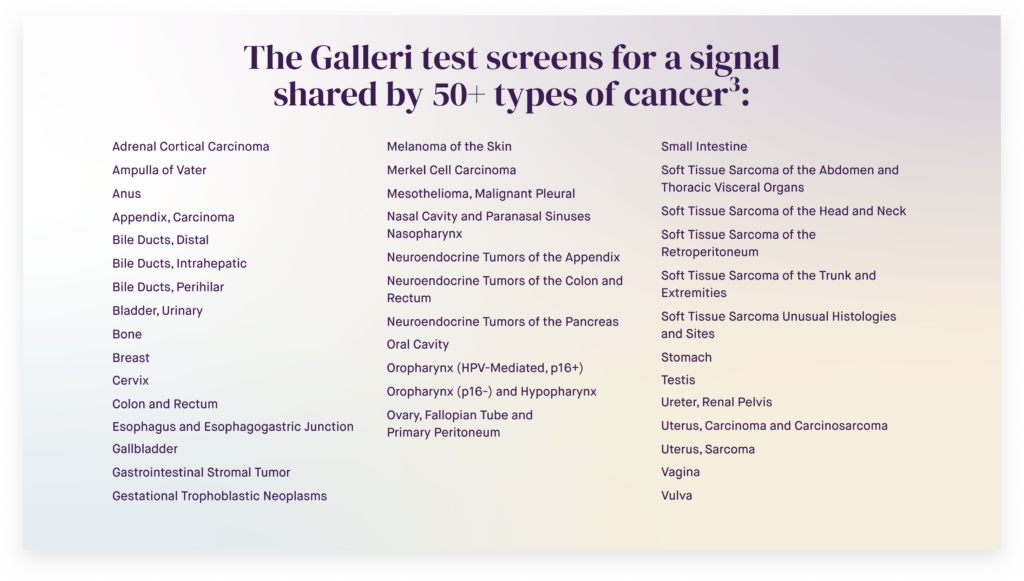
Cancer screening saves lives, but 70% of cancer deaths are caused by cancers without recommended screening tests.*1,2 The Galleri multi-cancer early detection (MCED) test can help find these cancers. With a simple blood draw, Galleri can detect more than 50 types of cancer before symptoms appear — when they can be easier to treat and are potentially curable.3
The Galleri test does not detect a signal for all cancers and not all cancers can be detected in the blood. False positive and false negative results do occur.
The Galleri test increases the number of cancers detected seven-fold when added to standard of care screening for breast, cervical, colorectal, and lung cancers and has the lowest false positive rate of any MCED test**.3,4,5,6
The test is especially sensitive to many of the deadliest cancers that are typically caught too late — like pancreatic, liver, and ovarian*** — and about half of Galleri-detected cancers are found at an early stage.3,6
The Galleri test identifies DNA in the bloodstream shed by cancer cells and does not predict future genetic risk for cancer.
The Galleri test delivers results you can trust
People who don’t have cancer have a very low likelihood of receiving a Cancer Signal Detected test result with Galleri. Galleri has a false positive rate of just 0.4%— the lowest of any available MCED test ** — helping to minimize unnecessary procedures and exposure to radiation.3,5,6
People who receive a Cancer Signal Detected test result with Galleri have a high likelihood of having cancer (positive predictive value of approximately 62%).3,4,6
Galleri is a screening test and does not diagnose cancer. Diagnostic testing is needed to confirm cancer. The Galleri test does not detect a signal for all cancers and not all cancers can be detected in the blood. False positive and false negative results do occur.
The Galleri test predicts a cancer signal of origin with 93.4% accuracy, guiding an efficient diagnostic work-up.6,7,8
Galleri screening is a game changer for cancer detection, helping adults at elevated risk of cancer, such as those 50 and over, detect aggressive cancers earlier and giving them more control over their health.
Rigorously tested
Published in top-tier peer-reviewed journals such as the Lancet and Annals of Oncology, Galleri has been rigorously studied with demonstrated performance in case-controlled and interventional studies, as well as extensive real-world experience.
Based on case-controlled and interventional intended use studies:
Galleri’s ability to correctly identify individuals who actually have cancer (sensitivity) is over 70% for the 12 cancers responsible for two-thirds of U.S. cancer deaths.3,4


How the Galleri test works
Clues in the blood: Cancer can stay hidden in the body for months or years, but many cancers leave clues in the blood — in the form of DNA released by the cancer cells — long before they cause symptoms.6,8
The Galleri test detects DNA fragments circulating in the blood and uses a machine learning algorithm to determine whether those DNA fragments originate from healthy cells or cancer cells, and for the ~1% of patients that receive a Cancer Signal Detected result, predict the most likely origin of the cancer. Galleri can identify DNA shed by cancer cells while the patient is still asymptomatic, when it may be more easily treated and potentially curable.
The Galleri test identifies DNA in the bloodstream shed by cancer cells and does not predict future genetic risk for cancer.
Two principles of biology behind the Galleri test: DNA shedding & methylation
DNA shedding is the natural process by which cells in the body shed small amounts of DNA into the bloodstream. Cancerous cells, if present in the body, also release DNA and so the blood may carry a mixture of DNA fragments from healthy and diseased or cancerous cells. To determine if each DNA fragment in the blood came from a cancerous cell or a healthy cell, we analyze methylation patterns.
Methylation is a natural process by which methyl groups are added to the DNA molecule. Methylation can change the activity of a DNA segment without changing the sequence by controlling which sections of DNA turn on or off. Typically, methylation turns genes “off” and demethylation turns genes “on.” GRAIL uses advanced machine learning algorithms and pattern recognition to “read” methylation patterns.
Using these two principles, the Galleri test has three scientific steps:
- Extract and sequence the DNA fragments from the blood to “read” the methylation patterns (assay)
- Determine if DNA fragments may have originated from cancerous cells (machine learning classifier)
- If cancerous DNA fragments are identified, use pattern recognition to predict the organ associated with the DNA fragments (cancer signal origin prediction, or CSO).
Population-level paradigm shift. Routine use of Galleri alongside recommended screening could decrease late-stage cancer diagnoses and usher in a new era of proactive cancer detection.
Available now, by prescription only.
*Assumes screening is available for all prostate, breast, cervical, and colorectal cancer cases and 43% of lung cancer cases (based on the estimated proportion of lung cancers that occur in screen-eligible individuals older than 40 years)
**Test performance metrics do not represent results of a head-to-head comparative study. Separate studies have different designs, objectives, and participant populations, which limits the ability to draw conclusions about comparative performance.
***The overall sensitivity in study participants with pancreatic cancer was 83.7% (61.9% for stage I, 60.0% stage II, 85.7% stage III, 95.9% stage IV). The overall sensitivity in study participants with liver/bile duct cancer was 93.5% (100% for stage I, 70.0% stage II, 100% stage III, 100% stage IV). The overall sensitivity in study participants with ovarian cancer was 83.1% (50.0% for stage I, 80.0% stage II, 87.1% stage III, 94.7% stage IV).[3]

Important Safety Information
The Galleri test is recommended for use in adults with an elevated risk for cancer, such as those age 50 or older. The test does not detect all cancers and should be used in addition to routine cancer screening tests recommended by a healthcare provider. The Galleri test is intended to detect cancer signals and predict where in the body the cancer signal is located. Use of the test is not recommended in individuals who are pregnant, 21 years old or younger, or undergoing active cancer treatment. Results should be interpreted by a healthcare provider in the context of medical history, clinical signs and symptoms. A test result of No Cancer Signal Detected does not rule out cancer. A test result of Cancer Signal Detected requires confirmatory diagnostic evaluation by medically established procedures (e.g. imaging) to confirm cancer. If cancer is not confirmed with further testing, it could mean that cancer is not present or testing was insufficient to detect cancer, including due to the cancer being located in a different part of the body. False positive (a cancer signal detected when cancer is not present) and false negative (a cancer signal not detected when cancer is present) test results do occur. Rx only.
Laboratory/test information
The GRAIL clinical laboratory is certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA) and accredited by the College of American Pathologists. The Galleri test was developed—and its performance characteristics were determined—by GRAIL. The Galleri test has not been cleared or approved by the Food and Drug Administration. The GRAIL clinical laboratory is regulated under CLIA to perform high-complexity testing. The Galleri test is intended for clinical purposes.
References
1. American Cancer Society. Cancer Facts & Figures 2022. https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2022.html 2. US Preventive Services Task Force. A,B,C grade recommendations, cancer, screenings. [cited 2023 Oct 23]. https://www.uspreventiveservicestaskforce.org/uspstf/topic_search_results 3. Klein EA, Richards D, Cohn A, et al. Clinical validation of a targeted methylation-based multi-cancer early detection test using an independent validation set. Ann Oncol. 2021 Sep;32(9):1167-77. doi: 10.1016/j.annonc.2021.05.806 4. Nabavizadeh N, et al. Safety and Performance of a Multi-Cancer Early Detection (MCED) Test in an Intended-Use Population: Initial Results from the Registrational PATHFINDER 2 Study. Proffered Presentation Presented at: European Society for Medical Oncology (ESMO) Annual Meeting; October 17-21, 2025; Berlin, Germany. 5. GRAIL, Inc. False positive rate. [Data on file: GR-2025-0256] 6. Schrag D, Beer TM, McDonnell CH, et al. Blood-based tests for multi-cancer early detection (PATHFINDER): a prospective cohort study. Lancet. 2023;402:1251-1260. doi: 10.1016/S0140-6736(23)01700-2 7. GRAIL, Inc. Enhanced Cancer Signal Origin prediction. [Data on file: VV-TMF-59592] 8. Hackshaw A, et al. Cancer Cell. 2022;40(2):109-13.